Qual o volume ocupado, a 2 atm de pressão, por…
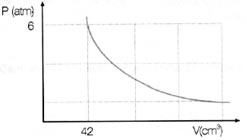
(PUC-PR) Qual o volume ocupado, a 2 atm de pressão, por certa massa de gás ideal que sofre transformações isotérmicas conforme o gráfico?
a) 42 cm3
b) 14 cm3
c) 21 cm3
d) 126 cm3
e) É impossível calcular, pois faltam dados.
Resolução
De acordo com o gráfico, o gás ocupa 42 cm3 (Vi) quando a pressão é igual a 6 atm (Pi). Dessa forma, para calcular o volume do gás (Vf) a 2 atm (Pf), basta utilizarmos a lei de Boyle:
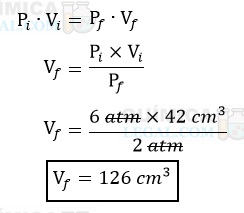
Gabarito: LETRA D
Confira mais EXERCÍCIOS RESOLVIDOS sobre ESTUDO DOS GASES.
Tá afim de ver mais RESOLUÇÕES COMENTADAS da PUC?



